Application
Biotechnology
Electroporation is widely used in microbial biotechnology due to its ability to induce reversible or irreversible permeabilization of bacterial, archaeal, and fungal cell membranes.
Applications in biotechnology
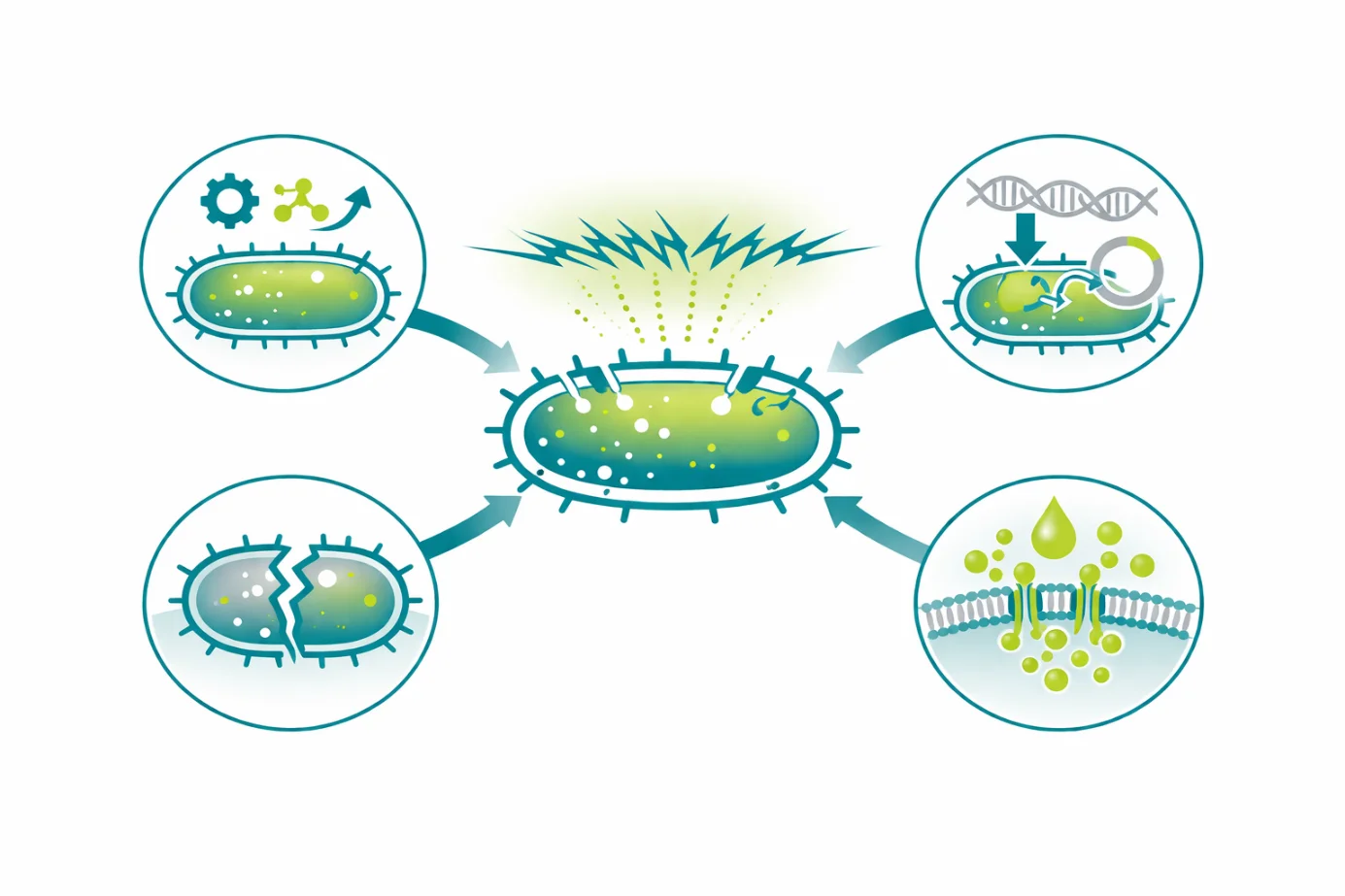
Electroporation is widely used in microbial biotechnology due to its ability to induce reversible or irreversible permeabilization of bacterial, archaeal, and fungal cell membranes. Key applications include genetic engineering of microorganisms, intracellular biomolecule extraction, microbial inactivation, and fundamental microbial research.
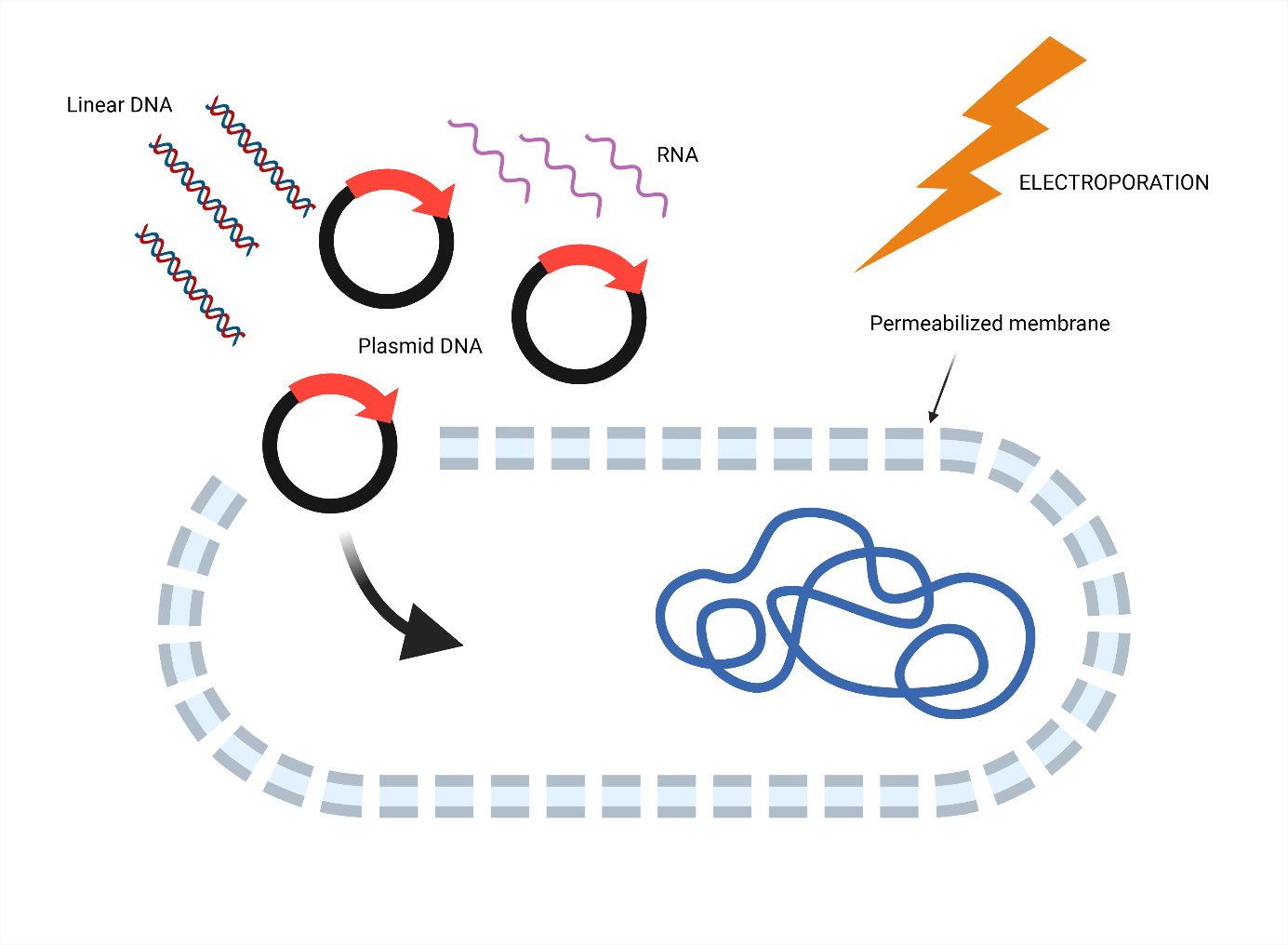
Genetic engineering of microorganisms
Electroporation is a key tool for microbial genetic engineering, enabling the introduction of plasmid DNA, linear DNA fragments, RNA, or genome-editing systems (e.g., CRISPR–Cas) into bacteria and yeast via reversible electroporation, during which transient pores are formed in the cell membrane (Figure 2) 2. This approach facilitates the development of recombinant and engineered strains for diverse industrial, medical, and environmental applications. Additionally, electroporation provides a controlled system for studying DNA uptake mechanisms and membrane permeability, offering insights into horizontal gene transfer 3 and microbial physiology.
Intracellular biomolecule extraction
Conventional biomolecule extraction methods typically rely on mechanical, chemical, or enzymatic disruption of microbial cell membranes, often resulting in extensive cell lysis and the concomitant release of intracellular contaminants such as host genomic DNA, lipopolysaccharides, and other endotoxins (Figure 3). In contrast, electroporation-mediated extraction enables the controlled and selective permeabilization of the cell membrane, allowing targeted release from microorganisms (e.g. bacteria, yeast, microalgae) of intracellular biomolecules (e.g. proteins, plasmid DNA, lipids, pigments) while preserving overall cellular integrity 4. This approach minimizes cell debris formation and significantly reduces contamination of the extracted product with undesired cellular components 5.
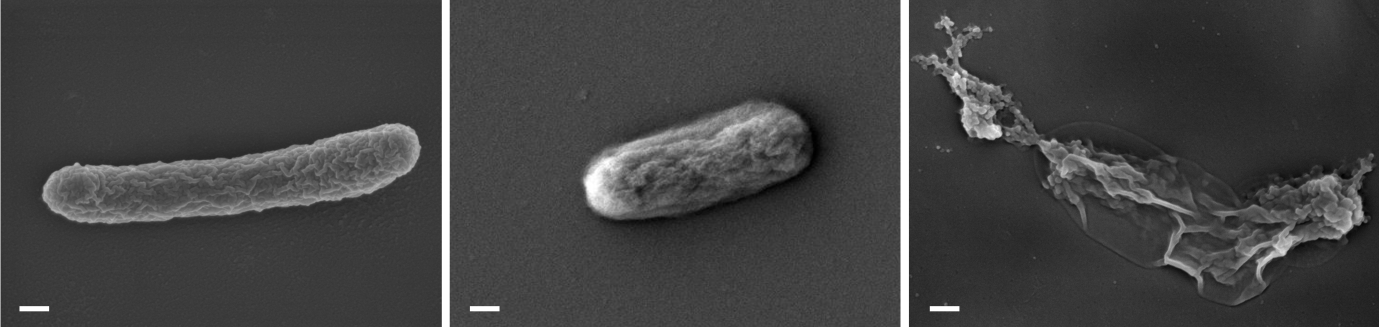
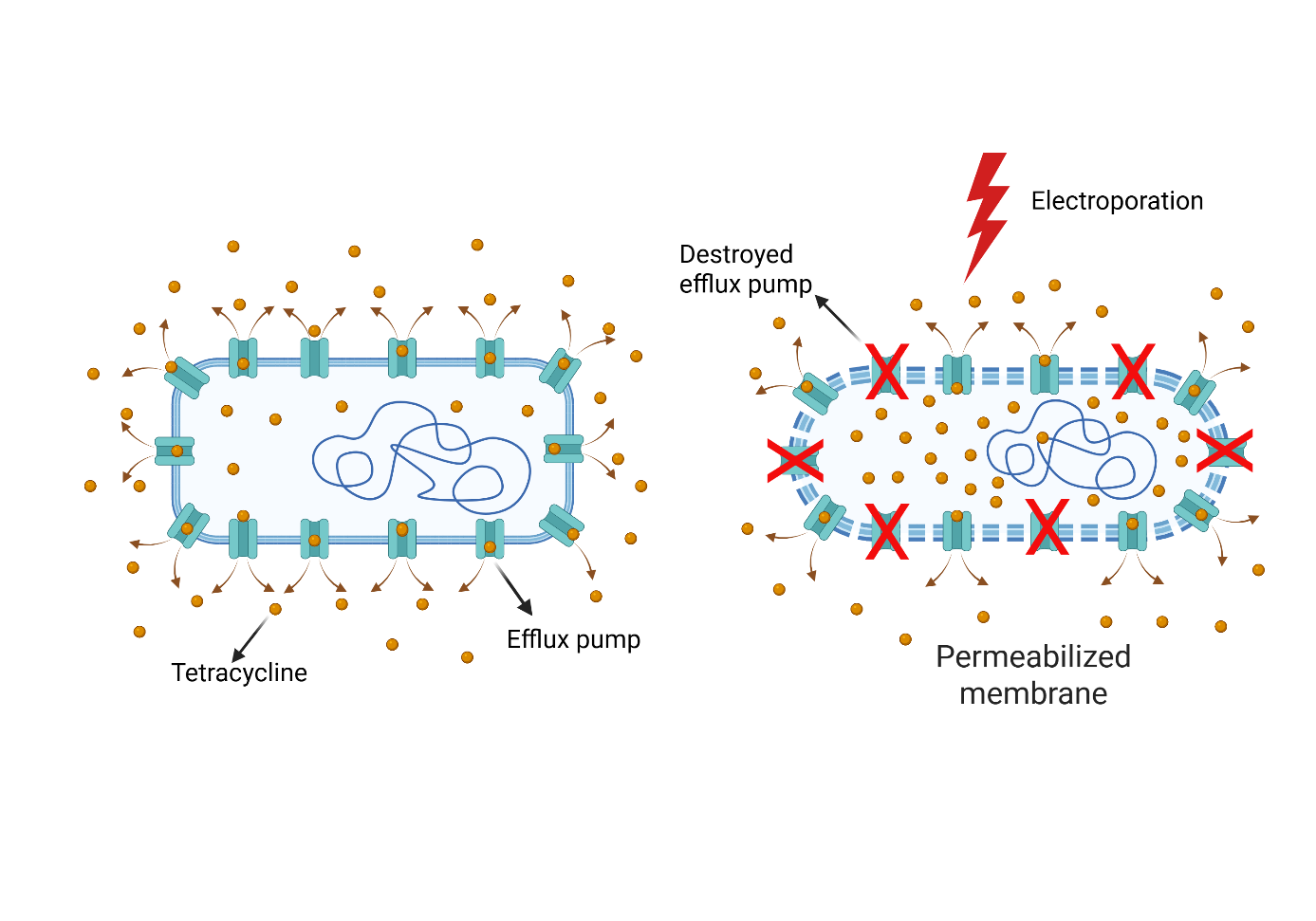
Microbial inactivation
Antibiotic-resistant bacterial infections represent an escalating challenge within contemporary healthcare systems. Despite the longstanding effectiveness of antibiotics in combating bacterial pathogens, their widespread overuse and misuse—particularly within the livestock industry—coupled with inadequate infection control practices, have significantly contributed to the emergence and spread of resistant bacterial strains 7. Accordingly, the development of effective strategies to mitigate antibiotic-resistant bacteria in wastewater is of critical importance. Our laboratory addresses this need by implementing a combined treatment strategy that employs both antibiotics and electroporation to inactivate resistant bacteria 8 (Figure 4). Electroporation has emerged as a highly promising adjunct technology, as it is effective against a broad spectrum of bacterial species, non-chemical in nature, and highly amenable to optimization.
Fundamental microbial research
Electroporation profoundly influences bacterial cells by modulating their physiology, membrane characteristics, membrane potential, and permeability, as well as influencing metabolic activity and motility. It can enhance growth, biomolecule production, and cell organization. Harnessing these effects enables the precise tailoring of bacterial behaviour for diverse applications, including improved fermentation efficiency, extended cell viability, and controlled adhesion in biotechnology and pharmaceutical processes 9.
 Tadej KotnikAntimicrobial EP
Tadej KotnikAntimicrobial EP Saša Haberl MegličMicrobial EP
Saša Haberl MegličMicrobial EP