Research approach
Numerical Modelling
Numerical modeling is a computational approach used to simulate physical processes by solving the underlying mathematical equations across multiple spatial scales.
Abstract
Numerical modeling is a computational approach used to simulate physical processes by solving the underlying mathematical equations. These equations are often impossible to solve analytically, especially in complex biological geometries. Instead, numerical solutions are obtained using a range of computational techniques – from atomistic, particle-based methods to the finite element method.
In our laboratory, we develop and apply such models to study electroporation across multiple spatial scales – from individual molecules to patient-specific organ geometries.
At the molecular level, we use molecular dynamics simulations to investigate how pores nucleate, grow, and reseal within the lipid bilayers and membrane proteins under applied electric fields, thereby providing nanoscale mechanistic insight that cannot be obtained experimentally.
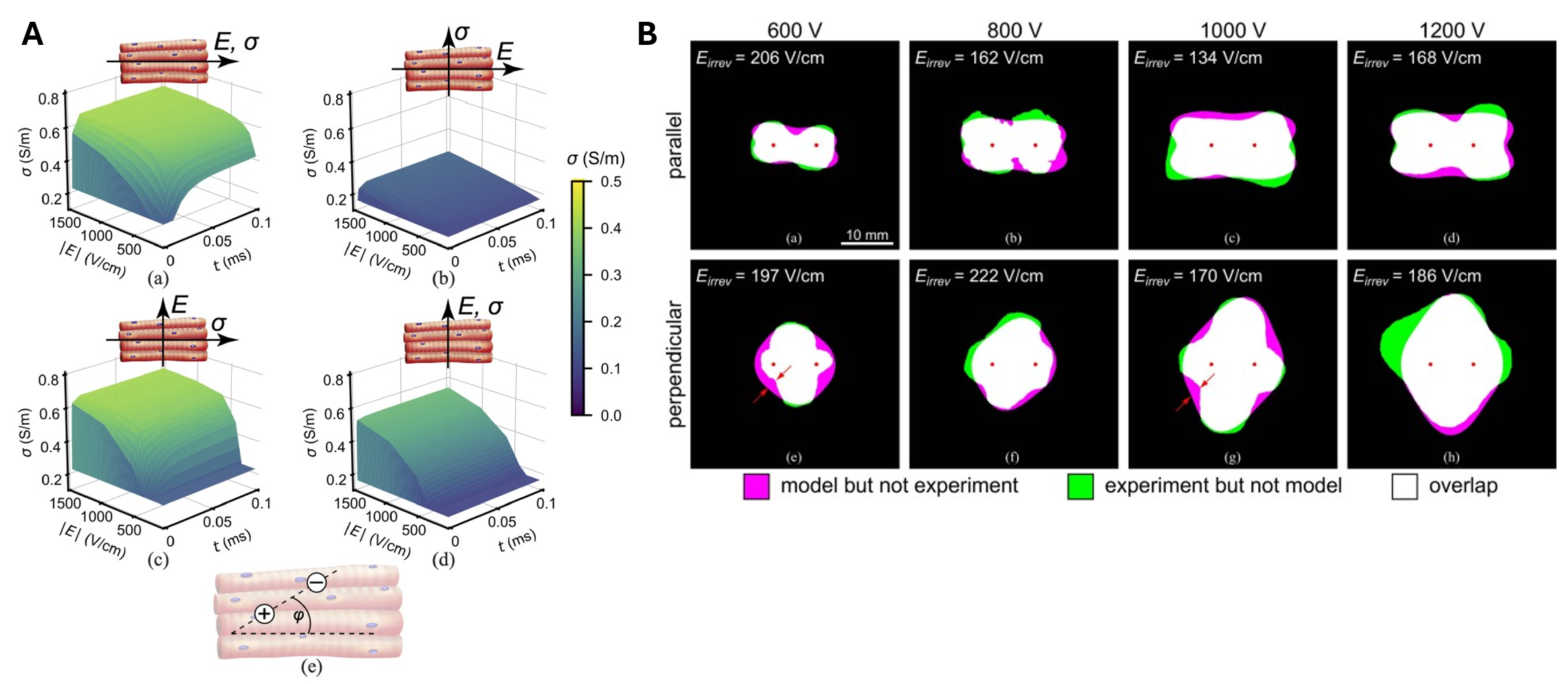
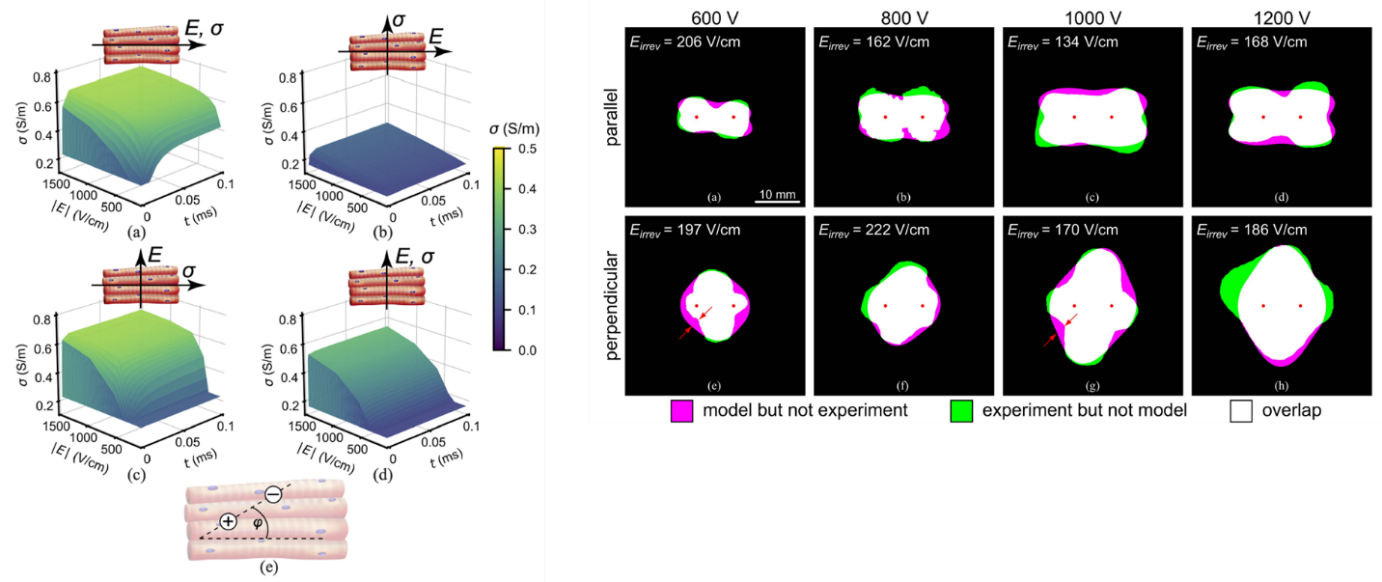
At the cellular level, we build models that describe transmembrane voltage induction, pore formation dynamics, and changes in membrane conductivity and permeability, accounting for realistic cell shapes such as cardiomyocytes, intracellular structures, and cell-to-cell interactions in dense tissue assemblies.
At the tissue level, we develop three-dimensional finite element models that couple the electric field distribution with bioheat transfer, incorporating tissue anisotropy (e.g., skeletal muscle and myocardial fiber orientation), inhomogeneities, and the nonlinear feedback between electric field, conductivity, and Joule heating that governs the evolving tissue response during electroporation.
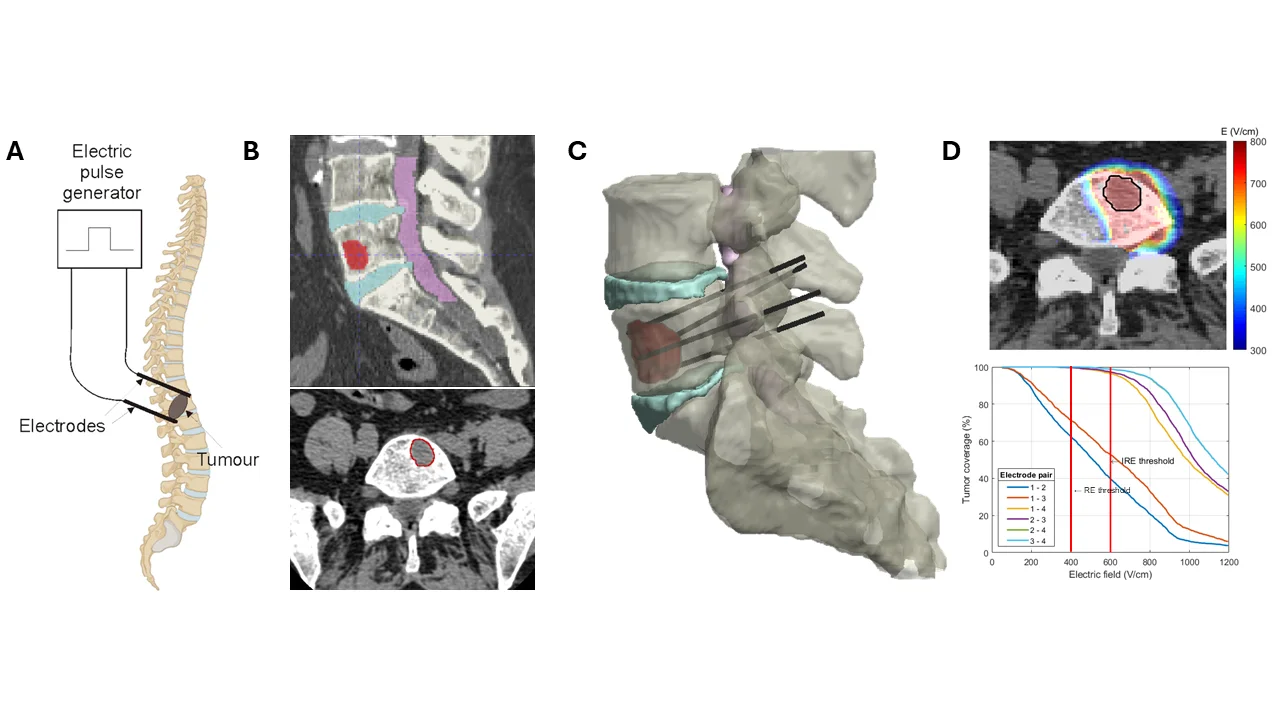
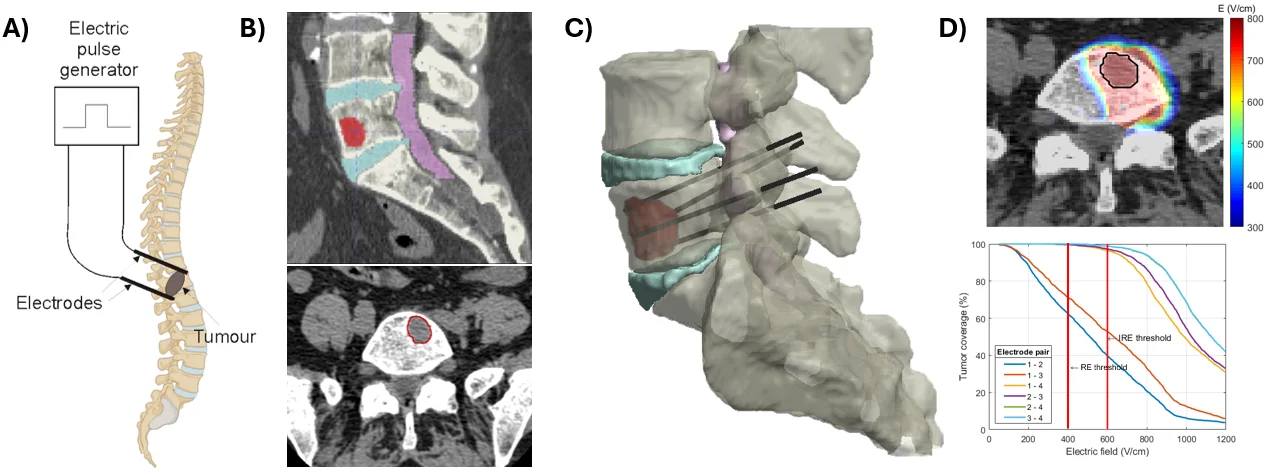
A central focus of our work is the construction of patient-specific models from clinical CT and MRI data. These anatomically accurate models serve two main purposes: investigating the safety and efficacy of emerging clinical applications, particularly pulsed field ablation for cardiac arrhythmias, and supporting computer-assisted treatment planning for electrochemotherapy and irreversible electroporation ablation of deep-seated tumors, where model-based optimization of electrode placement and pulse parameters is essential for achieving complete target coverage while sparing surrounding critical structures.
Electroporation from molecular dynamics perspective
Molecular dynamics (MD) simulations model electroporation at the atomistic scale by explicitly representing lipids, water, and ions in the cell membrane under an applied electric field. In electroporation, MD allows us to investigate the earliest membrane events that cannot be resolved experimentally – how pores nucleate, grow, stabilize, and reseal; how membrane composition and embedded proteins influence these pathways; and how electroporation alters membrane structure, permeability, and local transport. By providing mechanistic insight and quantitative parameters at nanometer and nanosecond scales, MD complements continuum and finite element models and helps connect molecular membrane behavior to cell- and tissue-level electroporation outcomes.
Modeling electroporation on the cellular level
On the subcellular and cellular levels, models describe the induction of transmembrane voltage, the initiation and dynamics of pore formation in the cell membrane, changes in membrane conductivity and permeability. We also consider the influence of cell shape, orientation, and intracellular structures on the electroporation outcome (see Figure 2). These models provide a mechanistic link between the applied electric field and membrane permeabilization, cell survival, or death. At an intermediate scale models consider assemblies of closely packed cells, capturing collective effects such as cell-to-cell interactions, volume fraction, and microstructural organization that cannot be represented by isolated single-cell models but are still below the homogenized tissue description.
Modeling electroporation on the tissue level
On the tissue level, electroporation is typically modeled in a three-dimensional anatomical domain by solving the partial differential equation for electric potential and deriving the electric field distribution using the finite element method (FEM) to accommodate realistic geometries, boundary conditions, and electrode configurations.
Simulations can be further extended with bioheat transfer and fluid flow dynamics to quantify Joule heating, perfusion-driven cooling, and, when relevant, convective transport by blood or interstitial fluids–effects that influence both safety margins and the final lesion pattern.
To reflect real tissue behavior, models incorporate bulk tissue properties (mechanical, electrical, and thermal), tissue inhomogeneities (such as multiple tissue layers or anatomical structures), and anisotropy where applicable (e.g., in skeletal muscle and myocardium, where fiber orientation strongly redirects current pathways).
During electroporation, tissue electrical conductivity is commonly modeled as a nonlinear function of the electric field, increasing due to the formation of conductive pathways in cell membranes. Simultaneously, Joule heating raises tissue temperature, which further increases conductivity through its temperature dependence. Together, these effects create nonlinear feedback between electric field, electrical conductivity, and heating that is captured in tissue-level FEM models to better describe the evolving tissue response to electroporation.
When combined with experimental lesion delineation, such models can be used to estimate lethal or reversible electric-field thresholds and related parameters (such as conductivity-increase factors) by fitting simulated field contours to measured lesion shapes, improving our ability to predict and interpret tissue response to electroporation.
Digital twins for clinical applications of electroporation
Patient-specific digital twins are built from medical images (CT or MRI), which are segmented to identify target tissues and relevant anatomical structures, and then used to construct three-dimensional, anatomically accurate models. Electrode or catheter geometries are introduced into these models, allowing tissue- and organ-level simulations that provide a macroscopic description of electroporation by computing the resulting electric field distribution in realistic patient anatomy. These models explicitly account for anatomical constraints, tissue inhomogeneities, and clinically feasible electrode placements, which are critical for predicting treatment outcomes in complex, deep-seated targets.
In our research, patient-specific digital twins are used for two main applications: (1) investigating the safety and efficacy of novel clinical applications, such as pulsed field ablation (PFA) for cardiac ablation, and (2) supporting patient-specific treatment planning for minimally invasive treatment of deep-seated tumors with electrochemotherapy (ECT) or irreversible electroporation (IRE) ablation.
Pulsed field ablation for treatment of cardiac arrhythmias
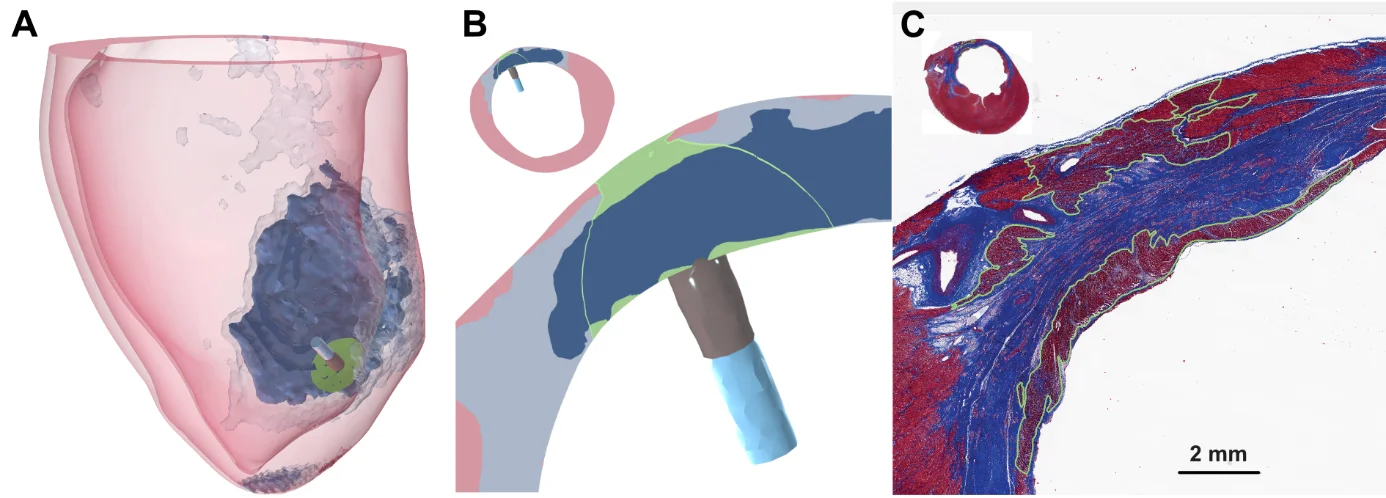
Pulsed field ablation (PFA) is an emerging non-thermal energy modality for treating cardiac arrhythmias by selectively destroying arrhythmogenic tissue through irreversible electroporation. We build patient-specific models of cardiac chambers from clinical CT and MRI data to simulate the electric field delivered by PFA catheters within realistic atrial and ventricular anatomies. These models incorporate myocardial fiber orientation, wall thickness variations, and the presence of pathological substrates such as scar and border zone tissue, enabling us to evaluate lesion transmurality, predict the extent of ablated tissue, and assess the safety of PFA with respect to surrounding structures such as the esophagus, coronary arteries, and phrenic nerve. Our simulations support both the mechanistic understanding of PFA lesion formation and the development of evidence-based dosing strategies for clinical use.
Computer-assisted treatment planning for tumor treatment
Tailored to each patient's anatomy, computer-assisted treatment planning aims to design a technically feasible plan that optimizes electrode placement and pulse parameters to ensure reliable coverage of the entire clinical target volume (e.g., a tumor with a safety margin) with an electric field exceeding the reversible electroporation threshold for ECT or the irreversible threshold for IRE ablation. At the same time, treatment planning aims to minimize unintended exposure and damage to surrounding healthy tissue and critical anatomical structures, while respecting anatomical and procedural constraints. By integrating patient-specific anatomy with numerical modeling of electric field distributions, treatment planning provides quantitative guidance for clinicians and supports the development of more precise, predictable, and optimized minimally invasive electroporation-based therapies, as illustrated on Figure 5 for percutaneous ECT of a spinal metastasis.
 Bor KosTreatment planning & cardiac PFA modelling
Bor KosTreatment planning & cardiac PFA modelling Lea RemsMolecular dynamics
Lea RemsMolecular dynamics Helena CindričTumor treatment planning
Helena CindričTumor treatment planning Rok ŠmercSkeletal muscle modelling
Rok ŠmercSkeletal muscle modelling Peter LombergarPEF chamber & tissue modelling
Peter LombergarPEF chamber & tissue modelling